Dissolved Oxygen Laboratory Report Essay Essay Example.
Conclusion. In conclusion, an increase in temperature and an increase in light increase the amount of dissolved oxygen within a water sample. Seeing that the results were consistent through multiple experiments, it is highly likely that if the experiment was repeated that similar or the same results would occur. There were two pieces.
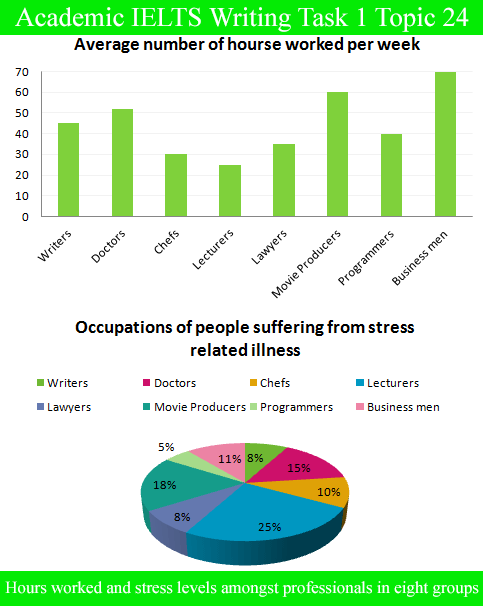
Dissolved Oxygen Laboratory Report Essay. 1. Based on the dissolved oxygen values for the three distilled water samples at different temperatures, it appears that there is a relationship between water temperature and DO concentration. It appears based on the averages of the class data that the lower the temperature of the water, the higher the.

Based on the dissolved O values for the three distilled H2O samples at different temperatures. it appears that there is a relationship between H2O temperature and DO concentration. It appears based on the norms of the category informations that the lower the temperature of the H2O. the higher the DO concentration. although merely to a certain point.

Based on the dissolved oxygen values for the three distilled water samples at different temperatures, it appears that there is a relationship between water temperature and DO concentration. It appears based on the averages of the class data that the lower the temperature of the water, the higher the DO concentration, although only to a certain point.

Phosphates and dissolved oxygen. - Phosphates are present in many natural waters, such as lakes and streams. Phosphates are essential to aquatic plant growth, but too much phosphate can lead to the growth of algae and results in an algae bloom. Too much algae can cause a decrease in the amount in dissolved oxygen in the water.

We also predict that all of our experimental variables will contain more dissolved oxygen than our control, the distilled water. Distilled water contains no organisms, is stagnant, and is kept at room temperature. These all contribute to a lower dissolved oxygen concentration.
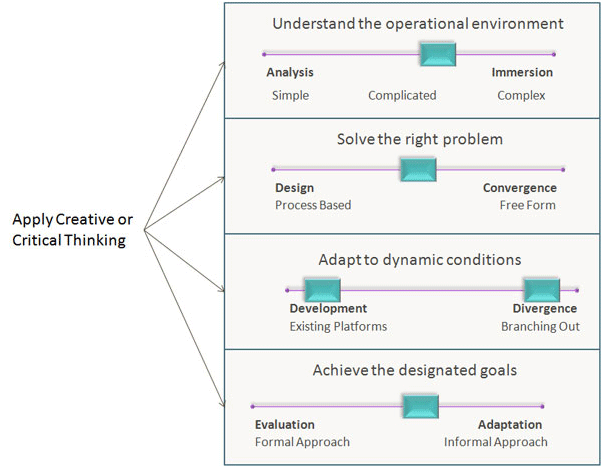
Under the condition of high temperature, oxygen gas forms weak molecular bonds with the water molecules. As a result, oxygen molecules will rise towards the surface and escape to the atmosphere, consequently reducing the amount of oxygen gas dissolved in the water.

Conclusion At the conclusion of the Dissolved Oxygen lab experiment, the K’ value from the equations listed previously helps to determine the correct solution that match the criteria put forth by the problem statement. Measurements were taken from the water flow through six PVC pipes.

Conclusion:-The Dissolved oxygen exists in water in a certain level. If any drop happens below this level the life forms in the water are unable to continue at a normal rate. It can cause killing of fishes and the growth of certain types of weeds which has a negative effect on the aquatic life.

Title: Pontoosuc Lake and Lake Stratification Abstract: The purpose of this lab was to examine the distribution of heat and dissolved oxygen throughout the water column from surface to bottom. We then will take this evidence and examine the profile for evidence of stratification or mixing.
More than 1000000 free essays. 2. Develop a hypothesis relating to the amount of dissolved oxygen measured in the water sample and the number of fish observed in the body of water.

Essay text: Oxygen is administered to patients whose breathing is impaired and also to people in aircraft flying at high altitudes, where the poor oxygen concentration cannot support normal respiration. Oxygen-enriched air is used in open-hearth furnaces for steel manufacture.

Pond Lab Report essays In the pond water lab experiment, our group has chosen to test the effects of nitrates on a pond community. After a lot of research, we have found that nitrates in a pond community can increase growth rates of plants and some animals. The effects of nitrates include algal bloo.